The Demineralized (DM) Water Plant Process: A Comprehensive Guide | Malaysia
Category: Wastewater Treatment
Specifications
Details
The Demineralized (DM) Water Plant Process: A Comprehensive Guide | Malaysia
Demineralized water (DM) is essential for various industrial applications, particularly in power plants, pharmaceuticals, electronics, and petrochemical industries in Malaysia. Given the growing emphasis on sustainable water management in industries such as semiconductor manufacturing, palm oil processing, and petrochemical refining, a well-designed DM Water Plant Process is crucial. This process effectively removes dissolved salts, minerals, and impurities from water, producing high-purity demineralized water with minimal conductivity and total dissolved solids (TDS).This guide provides an in-depth overview of the DM Water Plant Process, outlining each step, its significance, and the final water quality parameters. Additionally, we explore the importance of demineralized water in key Malaysian industries.
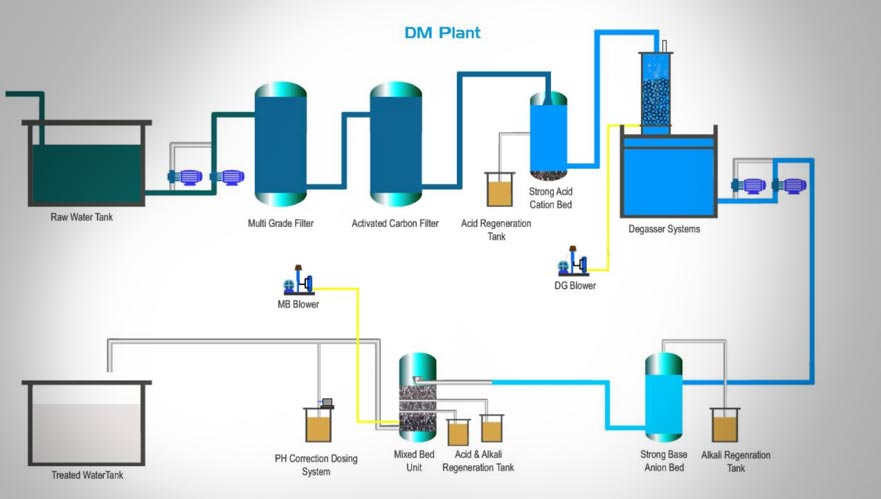
Step 1: Raw Water Storage
Raw water, which contains various dissolved impurities such as sodium (Na+), chloride (Cl-), calcium (Ca2+), and bicarbonate (HCO3-), is first collected in a Raw Water Tank. This storage ensures a consistent water supply for the treatment process. Raw water can originate from groundwater, rivers, lakes, or municipal sources, each with different impurity levels.Why is Raw Water Storage Important?
- Ensures a steady and controlled supply of water for treatment.
- Prevents pressure fluctuations that could affect downstream processes.
- Allows initial sedimentation of large particles, reducing the load on subsequent filtration steps.
Step 2: Multi-Grade Filtration (MGF)
After storage, raw water is passed through a Multi-Grade Filter (MGF) to remove suspended solids, dirt, and turbidity. This filtration system comprises multiple layers of filter media, including:- Graded Sand – Captures coarse particles.
- White Sand – Filters finer particulates.
- Fine Sand – Further polishes the water.
- Activated Carbon – Adsorbs chlorine, organic matter, color, and odor.
- Pebbles and Gravel – Support the filtration media and improve efficiency.
- Enhances the efficiency of downstream processes by reducing suspended solids.
- Protects delicate equipment from clogging and fouling.
- Reduces organic load, ensuring better performance of ion exchange resins.
Step 3: Activated Carbon Filtration (ACF)
The next step involves Activated Carbon Filtration, which removes organic contaminants, chlorine, and odor from the water. This process works on the principle of adsorption, where pollutants adhere to the porous surface of activated carbon granules.How Activated Carbon Works:
- Contaminants in water are attracted to and retained on the carbon surface.
- The highly porous structure of activated carbon increases adsorption capacity.
- Help in dechlorination, protecting ion exchange resins from oxidation.
- Removes chlorine, preventing damage to ion exchange resins.
- Eliminates taste, odor, and organic impurities.
- Reduces chemical contaminants like pesticides and volatile organic compounds (VOCs).
Step 4: Ion Exchange - Strong Base Cation Resin
After filtration, the water undergoes cation exchange using Strong Base Cation (SBC) Resin. This process removes positively charged ions (cations) such as calcium (Ca²⁺), magnesium (Mg²⁺), and sodium (Na⁺) by exchanging them with hydrogen ions (H⁺).Chemical Reactions Involved:
- Regeneration of Resin:
- HCl → H⁺ + Cl⁻
- R + HCl → RH⁺ + Cl⁻ (where R represents the resin)
- Ion Exchange Process:
- RH⁺ + Na⁺ → RNa⁺ + H⁺
- HCO₃⁻ + H⁺ → H₂CO₃ → H₂O + CO₂
Step 5: Degasser Tower
To remove carbon dioxide (CO₂), the water is passed through a Degasser Tower, ensuring stable demineralization and water purity.Step 6: Ion Exchange - Strong Base Anion Resin
Water undergoes anion exchange using Strong Base Anion (SBA) Resin, which removes negatively charged ions (anions) like chloride (Cl⁻), sulfate (SO₄²⁻), and nitrate (NO₃⁻).Step 7: Mixed Bed Resin Regeneration
To achieve the highest level of purity, water is further treated using a Mixed Bed (MB) Resin system, combining cation and anion exchange resins in a single unit.Why is Mixed Bed Resin Important?
- Achieves ultra-low conductivity levels.
- Removes residual ions, ensuring the highest purity standards.
- Essential for applications requiring ultrapure water, such as pharmaceuticals and semiconductor manufacturing.
The demineralized water produced from this process meets the following specifications:
- Total Dissolved Solids (TDS): 1-10 ppm
- pH Level: <7
- Conductivity: ≤0.2 μS/cm
The DM Water Plant Process is a highly effective method for producing high-purity deionized water. By integrating multi-stage filtration, ion exchange, degassing, and mixed bed resin polishing, this system ensures the removal of all dissolved impurities, making it suitable for various Malaysian industries such as semiconductors, petrochemicals, and power generation.
For more information on customized industrial water treatment solutions in Malaysia, contact us today to explore how we can help optimize your water treatment system!
View more about The Demineralized (DM) Water Plant Process: A Comprehensive Guide | Malaysia on main site


